This article was co-authored by Bess Ruff, MA. Bess Ruff is a Geography PhD student at Florida State University. She received her MA in Environmental Science and Management from the University of California, Santa Barbara in 2016. She has conducted survey work for marine spatial planning projects in the Caribbean and provided research support as a graduate fellow for the Sustainable Fisheries Group.
This article has been viewed 117,131 times.
With only basic kitchen supplies, you can make sodium acetate in your own home. This salt can be fun and practical to use. You can use sodium acetate to make ‘hot ice’ and/or hot ice sculptures. You can also put the sodium acetate into pouches for use as a reusable hand warmer. It is relatively simple and inexpensive to make, and only requires vinegar, baking soda, and some dishes.
Steps
Making a Sodium Acetate Solution
-
1Pour vinegar in a pan. Vinegar is a dilute solution of acetic acid. The solution is primarily water, with anywhere between 3% and 7% acetic acid. The acetic acid is a necessary ingredient in forming sodium acetate. Pour 500 millilitres (2.1 c) of vinegar into a pan.[1]
- Always wear safety goggles and gloves when handling acids and bases like vinegar and baking soda.
-
2Add baking soda to the pan. Baking soda is the common name for sodium bicarbonate. This ingredient will provide the sodium needed to form sodium acetate. Slowly sprinkle about 35 grams (7 tsp) of baking soda for every 500 millilitres (2.1 c) of vinegar.[2]Advertisement
-
3Stir the reaction. As you sprinkle the baking soda into the vinegar, you will see the solution start to bubble. This is due to the formation of carbon dioxide gas during the reaction. Use a stir rod or a spoon to keep the reaction moving, and prevent it from bubbling out of the container.[3]
- The reaction of vinegar with baking soda is as follows: NaHCO3 + CH3COOH ---> CH3COONa + CO2 + H2O
Boiling Off Excess Water
-
1Transfer the solution to a boiling pan. Any pan that is stove-safe will suffice. Transfer only the liquid solution. Do not pour solid baking soda into the boiling pan.
- You will only have solid baking soda if you added too much baking soda. The excess baking soda will remain in a solid (but wet) form.
-
2Bring the solution to a boil. Put the boiling pan on the stove and bring the solution to a slow boil. Avoid boiling vigorously, as this will make it hard to monitor the surface of the solution, and could lead to over-boiling. You can also use a bunsen burner or hotplate to boil the solution.[4]
-
3Watch the surface of the solution. Be sure that the solution is at a light enough boil that you can monitor the surface of the solution. If it is boiling too vigorously for you to watch the surface, turn the heat down. Allow the solution to slow boil until you see a white solid start to form in the solution or on the surface. When you see this, remove from heat immediately and swirl the solution until the solid redissolves.
-
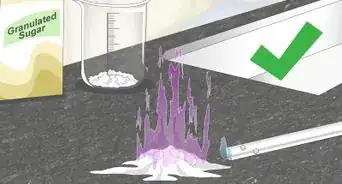
4Allow the solution to cool. As the solution cools, the sodium acetate dissolved in the hot water will precipitate out. It may take up to half an hour before you notice the formation of white sodium acetate crystals. Once the crystals form, you can pour off any excess water.[5]
- If the crystals do not form, the solution may be resting in a supersaturated state. This means that there is excess sodium acetate dissolved in the water for its current temperature. Drop a small piece of metal (even aluminum foil should work) into the solution to start the crystallization.
- If you are building hot ice sculptures, you will want to pour the solution a little at a time into your mold or design. This should catalyze the sodium acetate to precipitate out of solution, and form a solid sculpture.
-
5Scrape the crystals off. The crystals will form on the surface of the dish. For the best yield, scrape them off with a razor. Collect the crystals in an airtight container (a ziplock bag is sufficient).[6]
- If you plan to make a hand warmer, put the crystals in an airtight plastic bag. You can melt the crystals by dropping the bag into boiling water. Leave it in liquid form until you need a hand warmer, then drop a crystal or piece of metal in to catalyze the change back to a warm solid.
Evaporating Excess Water
-
1Pour the solution into an evaporating dish. An evaporating dish will allow the water and carbon dioxide to slowly evaporate from the crystals. This method will take much longer than boiling off excess water and carbon dioxide, but can be done. Do not transfer any solid baking soda particles into the evaporating dish.[7]
- A wide/long, shallow dish, like a glass casserole dish, works best. The water will take much longer to evaporate from a deep dish.
-
2Allow the contaminants to evaporate. The evaporation process will take days under standard conditions (room temperature, normal atmospheric pressure, etc.). If you wish to speed up the evaporation, you can place the dish under a heat lamp. As the water evaporates, white sodium acetate crystals will precipitate out of the solution and cling to the dish.
-
3Collect the crystals. Once the water evaporates, the sodium acetate crystals will be stuck to the evaporating dish. Use a razor to scrape the crystals from the dish. Store the crystals in an airtight container such as a ziplock bag.[8]
Expert Q&A
-
QuestionIs this safe for skin?
 Bess Ruff, MABess Ruff is a Geography PhD student at Florida State University. She received her MA in Environmental Science and Management from the University of California, Santa Barbara in 2016. She has conducted survey work for marine spatial planning projects in the Caribbean and provided research support as a graduate fellow for the Sustainable Fisheries Group.
Bess Ruff, MABess Ruff is a Geography PhD student at Florida State University. She received her MA in Environmental Science and Management from the University of California, Santa Barbara in 2016. She has conducted survey work for marine spatial planning projects in the Caribbean and provided research support as a graduate fellow for the Sustainable Fisheries Group.
Environmental Scientist Sodium acetate is relatively non-toxic; however prolonged contact with the skin can cause redness and irritation. Inhalation may cause a sore throat or couch so make sure you handle it in a well-ventilated area
Sodium acetate is relatively non-toxic; however prolonged contact with the skin can cause redness and irritation. Inhalation may cause a sore throat or couch so make sure you handle it in a well-ventilated area -
QuestionDoes this method make pure sodium acetate?
 Community AnswerNo, since supermarket vinegar usually contains impurities. If you want pure sodium acetate, you would need to start from very pure reactants.
Community AnswerNo, since supermarket vinegar usually contains impurities. If you want pure sodium acetate, you would need to start from very pure reactants. -
QuestionWhy should the salt be kept in an airtight place?
 CaeiiaTop AnswererMoisture from the air can eventually seep into a non-airtight place, making the salt moist and more likely to stick together. For whatever purpose you need this to be, wetness can also reduce the effect of this salt.
CaeiiaTop AnswererMoisture from the air can eventually seep into a non-airtight place, making the salt moist and more likely to stick together. For whatever purpose you need this to be, wetness can also reduce the effect of this salt.
Things You'll Need
Making Sodium Acetate Solution
- White distilled vinegar (Dilute 25%/5% Acetic Acid, C2H4O2+H2O)
- Baking soda (Sodium Bicarbonate, NaHCO3)
- Measuring cup
- Tablespoon
- Large bowl
- Gram Scale (Optional)
- Spoon
- Safety Goggles
Boiling Off Excess Water
- Small Saucepan
- Razor
- Airtight Container
- Heat Source
Evaporating Excess Water
- Evaporating Dish
- Heat Lamp (Optional)
References
- ↑ https://www.howtosmile.org/resource/making-sodium-acetate-hot-ice
- ↑ https://www.howtosmile.org/resource/making-sodium-acetate-hot-ice
- ↑ https://www.howtosmile.org/resource/making-sodium-acetate-hot-ice
- ↑ https://www.howtosmile.org/resource/making-sodium-acetate-hot-ice
- ↑ https://www.howtosmile.org/resource/making-sodium-acetate-hot-ice
- ↑ https://www.fleet.org.au/blog/hot-ice/
- ↑ https://www.fleet.org.au/blog/hot-ice/
- ↑ https://www.science-sparks.com/the-illusion-of-hot-ice/
About This Article
Before you make sodium acetate, put on safety goggles and gloves to protect yourself from the acids and bases involved in the process. Then, pour 500 milliliters of vinegar into a pan, sprinkle 35 grams of baking soda into it, and stir it together to keep the mixture from bubbling out of the container as the vinegar and baking soda react. Next, transfer the liquid mixture to a pan and bring it to a slow boil. After that, let it cool, pour off any excess water, and scrape the remaining sodium acetate crystals from the bottom of the pan. For tips from our Science reviewer on how to use evaporation to make sodium acetate crystals, keep reading!